
China Pharmaceutical Contract Manufacturing Market, By Service Type (Active Pharmaceutical Ingredient (API) Manufacturing, Finished Dosage Form (FDF) Manufacturing, Packaging & Labeling Services, Research & Development (R&D) Services, Drug Development & Manufacturing Services (CDMO)); By Product Type (Branded Drugs, Generic Drugs, Over-the-Counter (OTC) Drugs, Biologics & Biosimilars, Vaccines); By Dosage Form (Tablets, Capsules, Injectables, Syrups & Suspensions, Topical Formulations, Inhalers); By Therapeutic Area (Oncology, Cardiovascular Diseases, Infectious Diseases, Neurology, Respiratory Diseases, Diabetes, Others); By End User (Pharmaceutical Companies, Biotechnology Companies, Research Institutes); By Trend Analysis, Competitive Landscape & Forecast, 2021-2032
- Manufacturing
- Feb 2026
- Pages 200
- Report Format: pdf
- Report Price: $2500 USD
China Pharmaceutical Contract Manufacturing Market, Size & Forecast 2021-2032
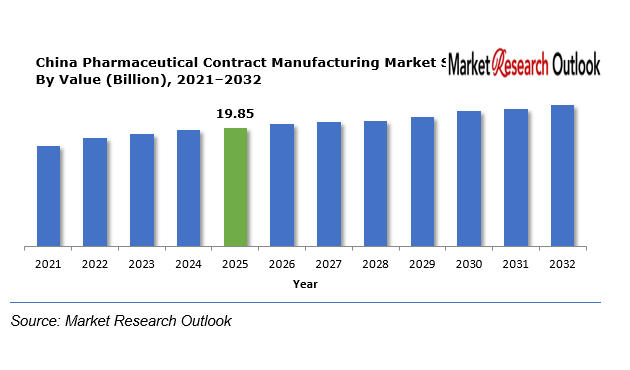
The China Pharmaceutical Contract Manufacturing Market size was estimated at USD 19.85 Billion in 2025. During the forecast period, the China Pharmaceutical Contract Manufacturing Market size is projected to grow at a CAGR of 11.4% reaching a value of USD 42.10 Billion by 2032. The market is expected to experience significant growth during the period from 2026 until 2032 because of the rising trend of global pharmaceutical manufacturing outsourcing and the robust domestic medicine demand and the fast development of biotechnology and biosimilar manufacturing. Pharmaceutical companies are turning to contract manufacturers for their production needs because this partnership helps them achieve cost-effective production and better manufacturing results and faster drug development times. Contract manufacturers are boosting their research capabilities and investing in modern manufacturing equipment to meet the rising demand for advanced therapies and biologics and complex generic medications.
Pharmaceutical Contract Manufacturing – Overview
Pharmaceutical contract manufacturing refers to the outsourcing of drug development and manufacturing and packaging and labeling services to specialized third-party manufacturers. The services enable pharmaceutical and biotechnology companies to produce their products at a lower cost while meeting regulatory and quality standards requirements. Contract manufacturers offer multiple services that include API production and finished dosage formulation and clinical trial material production and commercial-scale manufacturing.
China Pharmaceutical Contract Manufacturing Market
Growth Drivers
Increasing Global Outsourcing and Biopharmaceutical Expansion
The pharmaceutical manufacturing market in China experiences growth because companies increasingly outsource production to achieve cost savings and operational efficiency. Global pharmaceutical and biotechnology companies are increasingly partnering with Chinese contract manufacturers due to their large-scale production capabilities, advanced infrastructure, and competitive manufacturing costs. The increasing need for generic drugs and biosimilars and specialty medications has strengthened Chinese manufacturers position in the international pharmaceutical manufacturing network.
Challenges
Stringent Regulatory Compliance Requirements
The pharmaceutical contract manufacturing market in China faces difficulties because of changing regulatory standards and the requirement to uphold international quality certifications. The company needs to spend money on infrastructure and technological development because it must follow international Good Manufacturing Practices (GMP) and obtain regulatory approval from major markets which include the United States and Europe and maintain its quality control standards. The organization faces export limitations and product approval delays because it failed to meet established standards.
Geopolitical Impact on China Pharmaceutical Contract Manufacturing Market
The China Pharmaceutical Contract Manufacturing Market experiences its primary impacts through geopolitical factors which include global trade dynamics and regulatory changes and international supply chain realignments. Trade policies and export regulations together with tariffs establish the conditions which determine raw material availability and worldwide pharmaceutical product distribution. Multinational pharmaceutical companies base their outsourcing decisions on two factors which are international healthcare policies and changes in regulatory frameworks. Global pharmaceutical companies respond to geopolitical tensions by establishing multiple manufacturing sites which enable them to secure alternative supply routes. China continues as a primary center for pharmaceutical manufacturing because its strong infrastructure and technological capabilities and government support for the healthcare sector create an advantage.
China Pharmaceutical Contract Manufacturing Market
Segmental Coverage
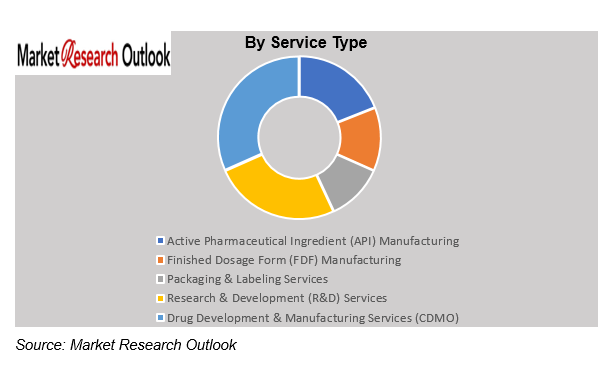
China Pharmaceutical Contract Manufacturing Market – By Service Type
Based on service type the China Pharmaceutical Contract Manufacturing Market is segmented into Active Pharmaceutical Ingredient (API) Manufacturing, Finished Dosage Form (FDF) Manufacturing, Packaging & Labeling Services, Research & Development (R&D) Services, and Drug Development & Manufacturing Services (CDMO). The API manufacturing segment will experience substantial expansion between 2026 and 2032 because of rising worldwide demand for both generic pharmaceuticals and specialty active pharmaceutical ingredients. Contract manufacturers expand their production capabilities while they invest in cutting-edge synthesis technologies to achieve international quality standards. Pharmaceutical companies will increasingly outsource their complete drug development and manufacturing operations which will drive growth in the CDMO and finished dosage form manufacturing sectors.
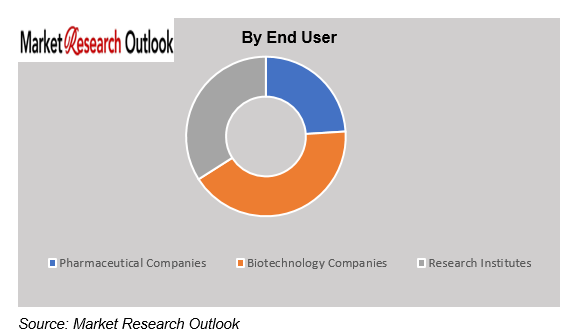
China Pharmaceutical Contract Manufacturing Market – By End User
Based on end user, the China Pharmaceutical Contract Manufacturing Market is segmented into Pharmaceutical Companies, Biotechnology Companies, and Research Institutes. The pharmaceutical companies segment will lead the market throughout the forecast period because companies will start outsourcing their manufacturing work to decrease their operational expenses and boost their operational performance. Pharmaceutical companies depend on contract manufacturers to produce their generic medications specialty pharmaceutical products and vaccines which enables them to supply escalating worldwide and national demand. Biotechnology companies and research institutes are establishing more partnerships with contract manufacturers to speed up their drug development and commercialization processes.
Competitive Landscape
Key participants in the China Pharmaceutical Contract Manufacturing market WuXi AppTec Co., Ltd., WuXi Biologics, Asymchem Laboratories (Tianjin) Co., Ltd., Porton Pharma Solutions Ltd., Joinn Laboratories (JOINN Biologics), Pharmaron Beijing Co., Ltd., Tigermed (Hangzhou Tigermed Consulting Co., Ltd.), Fosun Pharma (Shanghai Fosun Pharmaceutical Group), Hisun Pharmaceutical Co., Ltd., Shanghai RAAS Blood Products Co., Ltd., and other prominent players.
These companies are implementing strategic growth initiatives in order to gain a competitive advantage. The strategies being largely adopted include mergers and acquisitions, strategic alliances, joint ventures, licensing agreements, and new product launches. With the implementation of these strategies, the market participants aim to increase product portfolios, as well as enhance regional presence for long-term sustainable business growth in the Pharmaceutical Contract Manufacturing industry of China.
Scope of the Report
| Attributes | Details |
| Years Considered | Historical Data – 2021–2025
Base Year – 2025 Estimated Year – 2026 Forecast Period – 2026–2032 |
| Facts Covered | Revenue in USD Billion |
| Market Coverage | China |
| Product/ Service Segmentation | Service Type, Product Type, Dosage Form, Therapeutic Area, End User |
| Key Players | WuXi AppTec Co., Ltd., WuXi Biologics, Asymchem Laboratories (Tianjin) Co., Ltd., Porton Pharma Solutions Ltd., Joinn Laboratories (JOINN Biologics), Pharmaron Beijing Co., Ltd., Tigermed (Hangzhou Tigermed Consulting Co., Ltd.), Fosun Pharma (Shanghai Fosun Pharmaceutical Group), Hisun Pharmaceutical Co., Ltd., Shanghai RAAS Blood Products Co., Ltd., and other prominent players. |
Market Segmentation
- By Service Type
- Active Pharmaceutical Ingredient (API) Manufacturing
- Finished Dosage Form (FDF) Manufacturing
- Packaging & Labeling Services
- Research & Development (R&D) Services
- Drug Development & Manufacturing Services (CDMO)
- By Product Type
- Branded Drugs
- Generic Drugs
- Over-the-Counter (OTC) Drugs
- Biologics & Biosimilars
- Vaccines
- By Dosage Form
- Tablets
- Capsules
- Injectables
- Syrups & Suspensions
- Topical Formulations
- Inhalers
- By Therapeutic Area
- Oncology
- Cardiovascular Diseases
- Infectious Diseases
- Neurology
- Respiratory Diseases
- Diabetes
- Others
- By End User
- Pharmaceutical Companies
- Biotechnology Companies
- Research Institutes
- Research Framework
- Research Objective
- Product Overview
- Market Segmentation
- Executive Summary
- China Pharmaceutical Contract Manufacturing Market Insights
- Growth Drivers
- Restraints
- Opportunities
- Challenges
- Technological Advancements/Recent Developments
- Porter’s Five Forces Analysis
- Industry Value Chain & Entry Points
- China Pharmaceutical Contract Manufacturing Market: Regulatory Framework
- China Pharmaceutical Contract Manufacturing Market: Marketing Strategies
- China Pharmaceutical Contract Manufacturing Market Overview
- Market Size & Forecast, 2021-2032
- By Value (USD Billion)
- Market Share & Forecast
- By Service Type
- Active Pharmaceutical Ingredient (API) Manufacturing
- Finished Dosage Form (FDF) Manufacturing
- Packaging & Labeling Services
- Research & Development (R&D) Services
- Drug Development & Manufacturing Services (CDMO)
- By Product Type
- Branded Drugs
- Generic Drugs
- Over-the-Counter (OTC) Drugs
- Biologics & Biosimilars
- Vaccines
- By Dosage Form
- Tablets
- Capsules
- Injectables
- Syrups & Suspensions
- Topical Formulations
- Inhalers
- By Therapeutic Area
- Oncology
- Cardiovascular Diseases
- Infectious Diseases
- Neurology
- Respiratory Diseases
- Diabetes
- Others
- By End User
- Pharmaceutical Companies
- Biotechnology Companies
- Research Institutes
- By Service Type
- Market Size & Forecast, 2021-2032
- Demand Outlook & Customer Adoption Dynamics
- Demand Evolution By End-Use Industry
- Purchasing Behavior & Supplier Selection Criteria
- Demand Visibility & Contracting Trends
- Regional Demand Concentration & Customer Clusters
- Competitive Landscape
- List Of Key Players And Their Offerings
- China Pharmaceutical Contract Manufacturing Company Market Share Analysis, 2025
- Competitive Benchmarking, By Operating Parameters
- Key Strategic Development (Mergers, Acquisitions, Partnerships, Etc.)
- Geopolitical Impact On China Pharmaceutical Contract Manufacturing Market
- Company Profile
- WuXi AppTec Co., Ltd.
- Introduction & Company Profile
- Product Benchmarking
- Strategic Outlook
- Key Personals
- Key Competitors
- Financial Analysis
- Swot Analysis
- WuXi AppTec Co., Ltd.
**(Same Data Pointers Will Be Provided For The Below Companies)
- WuXi Biologics
- Asymchem Laboratories (Tianjin) Co., Ltd.
- Porton Pharma Solutions Ltd.
- Joinn Laboratories (JOINN Biologics)
- Pharmaron Beijing Co., Ltd.
- Tigermed (Hangzhou Tigermed Consulting Co., Ltd.)
- Fosun Pharma (Shanghai Fosun Pharmaceutical Group)
- Hisun Pharmaceutical Co., Ltd.
- Shanghai RAAS Blood Products Co., Ltd.
- Other Prominent Players
- Key Strategic Recommendations
- Research Methodology
- Qualitative Research
- Primary & Secondary Research
- Quantitative Research
- Market Breakdown & Data Triangulation
- Secondary Research
- Primary Research
- Breakdown Of Primary Research Respondents, By Country
- Assumption & Limitation
- Qualitative Research
* Financial information in case of non-listed companies will be provided as per availability
**The segmentation and the companies are subjected to modifications based on in-depth secondary for the final deliverable
